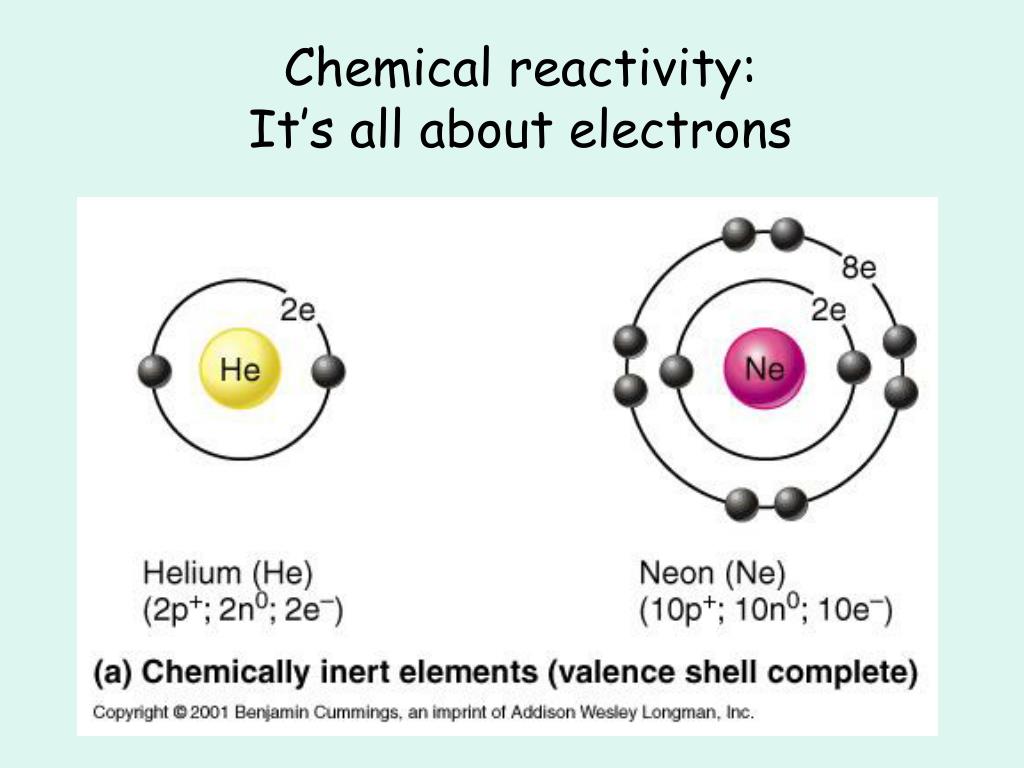
The existence of unfair DH electrons in the Vaillant shells. Of course, I'm anthropomorphizing Adams, but Adams or people Teo rate, so this really isn't going to have an impact on reactivity. 
So the average distance of the outermost electron shell from the nucleus isn't going to have a lot to do with what the Adam essentially wants, which is to have completed feeling shells. the average distance of the outermost electron shell from the nucleus. You want to have the outer most valence shell to be filled or to have no violence shows at all. The potential energy of the valence shell e. In general, protons are directly involved in the reactivity of peroxidases ( Scheme 1) and, as a consequence, ferryl centre protonation has been essential to justify the different FeO bond distances measured for compounds I and II by X-ray diffraction structural analysis 14. The sum of the potential energies of all the electron shells d. The existence of unpaired electrons in the valence shells c. In general, two classes can be distinguished: the transformation of the surface by a (real) chemical reaction, like e.g. The average distance of the outermost electron shell from the nucleus b. Almost all surface reactions involve the formation of a surface layer with a different lattice constant than the original surface. What I'm sure you've realized is that the reaction's primarily occur in order to have a it's have stable failings, shelves and those valence shells. The reactivity of an atom arises from Answer a. A difference in charge arises in different areas of the atom due to the uneven spacing of the electrons between the atoms. The reactivity of an Adam arises from a the average distance of the average of the outermost electron shell from the nucleus be the existence of unpaid electrons in the vale in shells sea, the sum of potential energies of all the electron shells or D, the potential energy of the violence shelves.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
